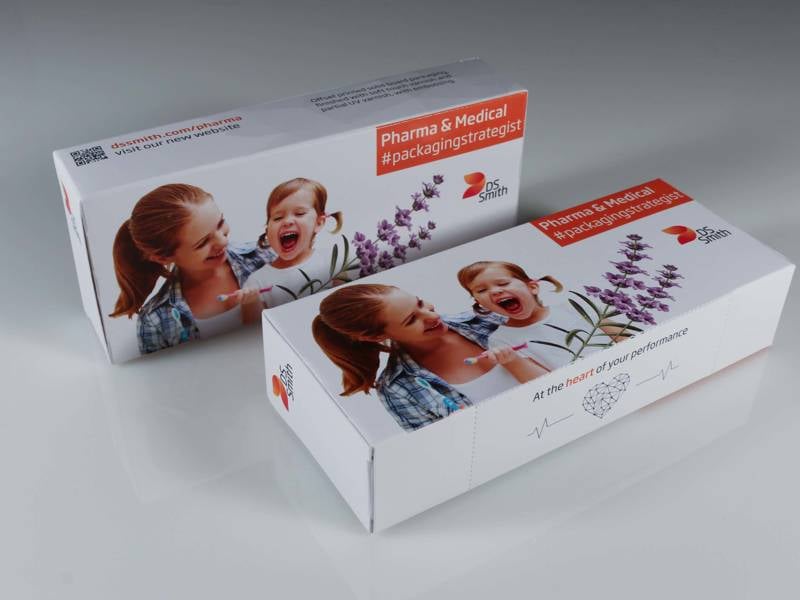
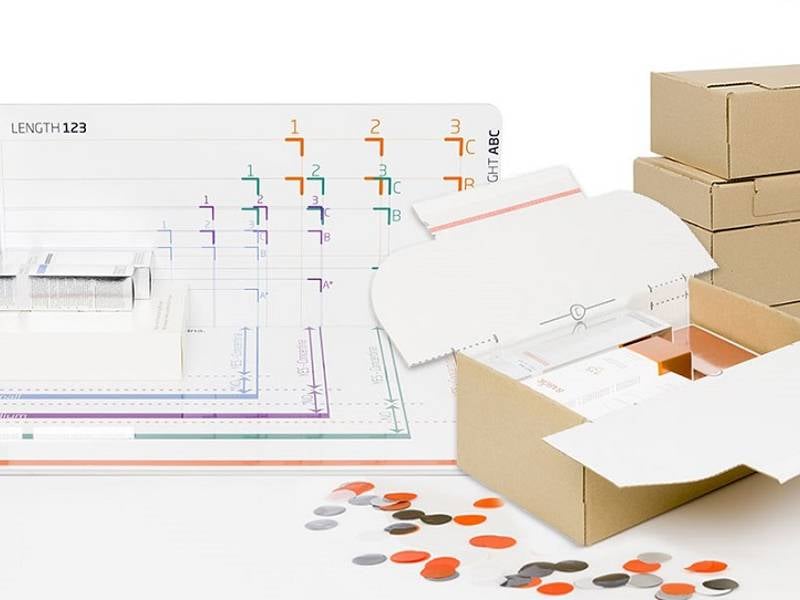
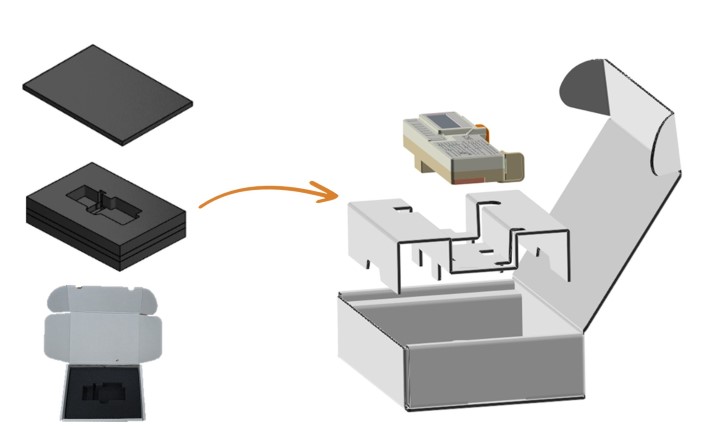
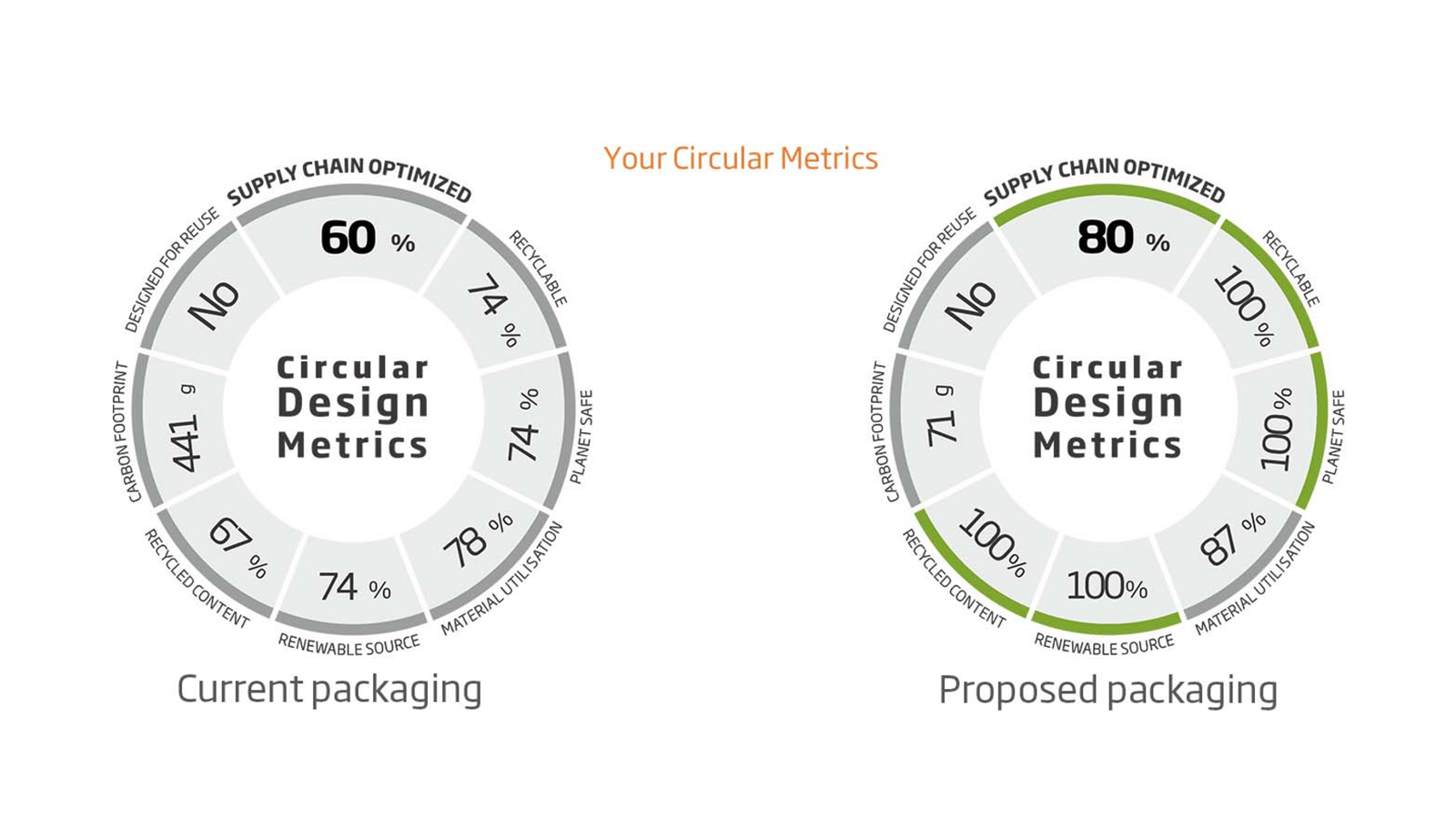
Dedicated medical and pharmaceutical packaging solutions
We understand the complexities and challenges of the pharmaceutical and medical markets. Our pharma packaging solutions are safe, sustainable, high quality and compliant with strict quality requirements.